Imidazopyridine
Federal government websites often end in, imidazopyridine. The site is secure. BoxUnited Arab Emirates. Fused pyridines are reported to display various pharmacological activities, imidazopyridine as antipyretic, analgesic, antiprotozoal, antibacterial, antitumor, antifungal, anti-inflammatory, and antiapoptotic.
Potent serine palmitoyl transferase inhibitor. We continue to work to improve your shopping experience and your feedback regarding this content is very important to us. Please use the form below to provide feedback related to the content on this product. By clicking Submit, you acknowledge that you may be contacted by Fisher Scientific in regards to the feedback you have provided in this form. We will not share your information for any other purposes.
Imidazopyridine
Imidazopyridine scaffold has gained tremendous importance over the past few decades. Imidazopyridines have been expeditiously used for the rationale design and development of novel synthetic analogs for various therapeutic disorders. A wide variety of imidazopyridine derivatives have been developed as potential anti-cancer, anti-diabetic, anti-tubercular, anti-microbial, anti-viral, anti-inflammatory, central nervous system CNS agents besides other chemotherapeutic agents. Imidazopyridine heterocyclic system acts as a key pharmacophore motif for the identification and optimization of lead structures to increase medicinal chemistry toolbox. The present review highlights the medicinal significances of imidazopyridines for their rationale development as lead molecules with improved therapeutic efficacies. This review further emphasis on the structure-activity relationships SARs of the various designed imidazopyridines to establish a relationship between the key structural features versus the biological activities. Communicated by Ramaswamy H. Keywords: Biological activity; imidazopyridine; leads; molecular modeling; structural activity relationship. Abstract Imidazopyridine scaffold has gained tremendous importance over the past few decades. Publication types Review.
Wu, imidazopyridine, W. On the importance of an acid additive in imidazopyridine synthesis of pyrido [1, 2-a] benzimidazoles by direct copper-catalyzed amination.
This moiety is also useful in material science because of its structural character. Synthesis of this moiety from the easily available chemicals is desirable due to its tremendous use in the various branches of chemistry. Here we report a review on the synthesis of this scaffold employing different strategies such as condensation, multicomponent reactions, oxidative coupling, tandem reactions, aminooxygenation, and hydroamination reactions. Bagdi, S. Santra, K. Monir and A. Hajra, Chem.
Federal government websites often end in. The site is secure. Imidazopyridines constitute one of the most important scaffolds in medicinal chemistry, as their skeleton could be found in a myriad of biologically active molecules. Although numerous strategies were elaborated for imidazopyridine preparation in the s, novel eco-compatible synthetic approaches have emerged, conscious of climate change concerns. In this framework, photochemical methods have been promoted to conceive this heterocyclic motif over the last decade. This review covers the recently published works on synthesizing highly functionalized imidazopyridines by light induction.
Imidazopyridine
An imidazopyridine is a nitrogen containing heterocycle that is also a class of drugs that contain this same chemical substructure. In general, they are GABA A receptor agonists , however recently proton pump inhibitors , aromatase inhibitors , NSAIDs and other classes of drugs in this class have been developed as well. Despite usually being similar to them in effect, they are not chemically related to benzodiazepines. As such, GABA A -agonizing imidazopyridines, pyrazolopyrimidines , and cyclopyrrones are sometimes grouped together and referred to as " nonbenzodiazepines. NSAIDs , analgesics and antimigraine drugs:. Drugs acting on the cardiovascular system :. Antineoplastic agents:. Contents move to sidebar hide.
Cbc listen live
Account Administration. Preliminary mechanistic studies indicate that this reaction proceeds through a catalytic Ortoleva-King reaction. Afzal B. Signatures of compounds according to Althagafi et al. Compound 6d showed two hydrogen bonds with Asp and Cys and the same pi-pi interactions with P Also, the clustering method was applied in the equilibrium time range by gromos approach and cut off value 0. Yamaoka, T. The target compounds 6a — 6l were synthesized as shown in Fig. Conversion of pyridine to imidazo [1, 2-a] pyridines by copper-catalyzed aerobic dehydrogenative cyclization with oxime esters. Shekar, R. Zhang, Q. The combination of flavin and iodine catalyzes an aerobic oxidative C-N bond-forming process for the facile synthesis of imidazo[1,2- a ]pyridines. Agarose 1.
This is a preview of subscription content, log in via an institution to check access.
Novel cyano-and amidino-substituted derivatives of styrylbenzimidazoles and benzimidazo [1, 2-a] quinolines. Moreover, 6d and 6f also inhibited the proliferation of cancer cells Table 2. All these derivatives exhibit a wide range of pharmacological activities, such as antipyretic, analgesic, anxiolytic, antiprotozoal, antibacterial, antitumor, antifungal, anti-inflammatory, and antiapoptotic activities [ 10 , 11 , 12 , 13 , 14 , 15 ]. Life Sciences Products. We further observed that when compounds 6d , 6e and 6f were incubated with AsPc-1 cells, the number of early and late apoptotic cells were significantly increased, while the number of live cells were decreased. Shop All Lab Furniture and Storage. Frett, H. Figure 2. Imidazopyridine scaffold has gained tremendous importance over the past few decades. In recent years, IZPs have been synthesized and screened for their in vitro and in vivo activity against Gram-negative, Gram-positive, and resistant strains of bacteria.

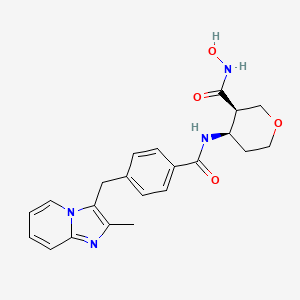
I am assured, that you are not right.
Please, more in detail